How SARS-CoV-2 Evades And Suppresses The Immune System (Part 5)
(Posted on Thursday, August 19, 2021)
This is the fifth article in a 15-part series called “Relevance of Immune Suppression by SARS-CoV-2 to Understanding and Controlling the Covid-19 Pandemic,” which will explore an underappreciated but highly significant aspect of SARS-CoV-2 replication. The ability of SARS-CoV-2 to delay, evade, and suppress the immune system has myriad implications for drugs, vaccines, and other aspects of our pandemic response. The first set of pieces in this series are intended for a general audience; the second set, for the medical community; and the third and final set, for biomedical researchers looking for a deeper understanding of variants, how they’re generated, and what we might do to control them. Read part one, part two, part three, and part four.

3D illustration of a besieged castle
GETTY
The second section of this series, which begins with this piece, will discuss language and an amount of detail suitable for the lay person with a serious interest in science and medicine.
If viral proteins were manufactured out in the open, they would run the risk of drawing attention from innate immune sensors suspended in the cytoplasm of the cell. To minimize risk, the virus sets itself up in a privileged compartment, a double membrane vesicle, which serves as a replication site where viral enzymes can whir away unnoticed. Like soldiers under siege in a well-armed fortress, so long as the structure holds, they will remain protected and out of sight.
These compartments are a recurring motif among viruses, including poliovirus, hepatitis C, and coronaviruses more broadly. Their biogenesis is not completely understood, but it is expected they derive from the membranes of well-worn cellular pathways that conduct routine functions for the cells, such as disposal of damaged cellular components. The formation of these is facilitated by the interaction of virus-specific proteins, which modify the formation of such vesicles to the virus’ advantage.
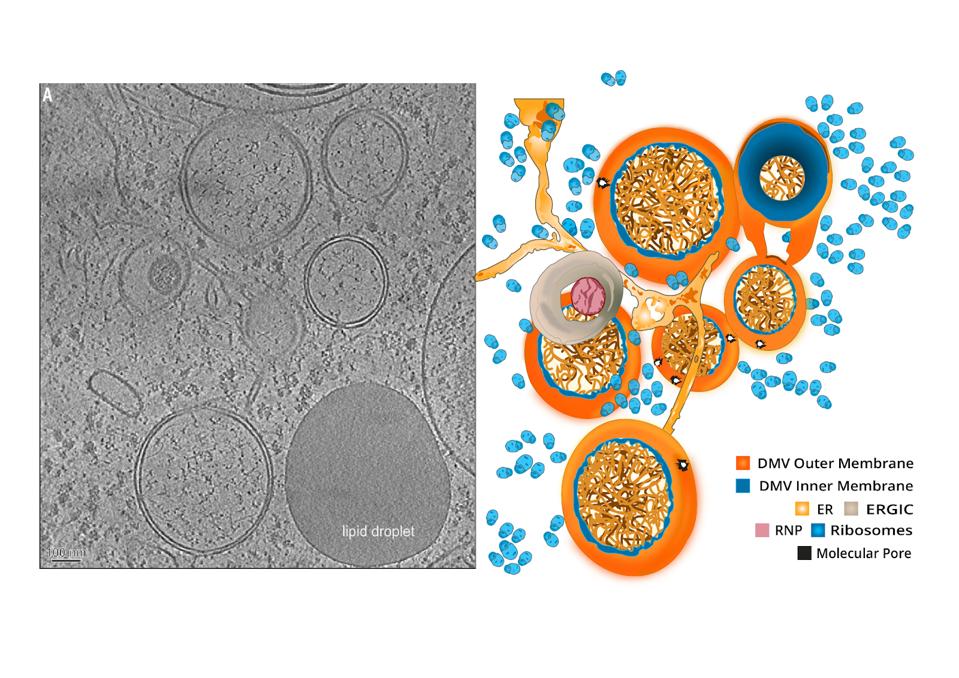
Coronavirus-induced DMVs revealed by cryo-ET.
“A MOLECULAR PORE SPANS THE DOUBLE MEMBRANE OF THE CORONAVIRUS REPLICATION ORGANELLE” HTTPS://SCIENCE.SCIENCEMAG.ORG/CONTENT/369/6509/1395A double membrane vesicle serves two main purposes. First, it concentrates viral proteins and host factors in one central location. The second I alluded to previously—it helps the virus obscure its replication intermediates from possible immunodetection. In particular the cell has a series of alarms that detect single and double stranded RNAs. Scecreting the secondary intermediates required for such a vesicle prevents detection by what are called toll receptors.
The outer membrane of the double membrane vesicle interconnects with the endoplasmic reticulum (ER) and other virus-induced membranes. For SARS-CoV it was suspected that replication sites were formed using the ER membrane, while budding sites also used membranes from the Golgi apparatus and endoplasmic-reticulum–Golgi intermediate compartment.
According to prior research on SARS-CoV and MERS-CoV, the nonstructural proteins NSP3 and NSP4 play a likely role in the formation of these privileged compartments. NSP6 and other host factors might be involved also. All three proteins have transmembrane domains that recruit the scaffolding of host cell membranes for their own purposes.
NSP3 has membrane disordering and proliferation abilities that allow it to either participate in membrane production or expand existing membranes. It contains two transmembrane regions that interact with ER membranes and cause membrane buildups in infected cells. The luminal loops of NSP3 and NSP4 interact with counterparts on the opposite side of the ER, inducing membrane pairing. NSP3 and NSP4 also associate with NSP6 during the assembly of the replication complex. Study of NSP6 in other viruses has established that it can induce autophagosomes, a particular type of double membrane vesicle that delivers cellular detritus to lysosomes for degradation. NSP6 might interfere with adaptive immunity by ensuring any immunomodulatory proteins end up in autophagosomes, thus halting their ability to counteract the invading pathogen.
Some viral replication organelles, like those seen in picornaviruses, have open configurations that accommodate easy mobility between the compartment and surrounding cytosol. In coronaviruses, they’re closed—which raises the question of how newly minted viral mRNAs are exported for translation.

Architecture of the molecular pores embedded in DMV membranes.
“A MOLECULAR PORE SPANS THE DOUBLE MEMBRANE OF THE CORONAVIRUS REPLICATION ORGANELLE” HTTPS://SCIENCE.SCIENCEMAG.ORG/CONTENT/369/6509/1395The solution, cryo-electron microscopy studies have identified, is a pore that spans the inner and outer membrane of the vesicle—a bridge connecting the cloistered interior to the surrounding cytosol. NSP3 is the primary building block behind this complex, though NSP4 and NSP6 are likely fellow constituents.
In the next piece for this series, I will discuss at length another element of the virus’ immune suppression strategy: mRNA mimicry.
Originally published on Forbes (August 19, 2021)

